A limiting factor is a resource or environmental condition which limits the growth, distribution or abundance of an organism or population within an ecosystem. Psychology definition for Between Subjects Design in normal everyday language, edited by psychologists, professors and leading students. Help us get better. Between Subjects Design. All material within this site is the property of AlleyDog.com.
MethodsSix non-holder subjects were studied on three occasions at 7–16 day intervals; doses (20–170 mg/day) remained unchanged. Blood samples and pharmacodynamic data were collected 10–12 times over a 24-h inter-dosing interval. All pharmacodynamic data were expressed as the area under the end-point versus time curve. Using analyses of variance with mixed effects, best estimates were made of the ratio of between- to within-subject variation, with corresponding 95% confidence intervals (CI) for within-subject variation at the average value. ResultsSubjects were relatively consistent between occasions, whereas there was much greater between-subject variability ( P. IntroductionMethadone is the drug most widely used as substitution treatment for opioid dependence.
Although it is universally administered as a racemate, the (R)-enantiomer is the pharmacologically active moiety and is more effective than the racemate in suppressing withdrawal ,. Its high oral bioavailability and long terminal half-life (average 48 h or greater , ) form the basis for the once-daily oral regimens that prevail in almost all methadone maintenance treatment (MMT) programmes worldwide.
The main objectives of successful substitution treatment are rehabilitation and socialization of the dependent individual, decrease in needle-associated diseases and mortality, decrease or elimination of illicit drug use and decrease in crime cost by the user and to the community. Randomized controlled trials of MMT programmes in different countries have shown them to largely fulfil these aims –. However, to achieve these outcomes successfully, MMT programmes need to be able to retain patients in treatment, often indefinitely.Even in programmes with liberal dosing policies, some patients experience withdrawal symptoms and decreased methadone efficacy for part of the dosing interval. We have shown that 34% of a nonselected sample of patients regularly experienced breakthrough withdrawal during the once daily interdosing interval.
These individuals were designated as nonholders, wheras the remainder were designated as holders. The nonholders also reported significantly fewer subjective (pleasurable) opioid effects than holders.Detailed pharmacodynamic/pharmacokinetic analyses of holders and nonholders over one dosing interval at steady state have shown that, in both groups, there is evidence of an inverse relationship between plasma racemic methadone concentrations and withdrawal severity and total mood disturbance , and a direct relationship between plasma concentrations and subjective opioid effects. Similar results have been reported for (R)-methadone ,. In addition, there is evidence of a very steep plasma concentration–effect relationship for withdrawal and positive opioid effects, with mean Hill slope factors of 5.4 and 5.1, respectively ,. This means that small changes in plasma methadone concentrations would translate into relatively large changes in response.Methadone, in common with many drugs, shows considerable between-individual variability in its pharmacokinetics and pharmacodynamics.
This has been reported for racemic methadone following single doses in pain patients and for (R)-methadone at steady state in MMT patients ,. This variability in pharmacokinetics has been regarded by some as limiting its more widespread use in pain treatment –. Methadone is highly bound to plasma α 1-acid glycoprotein and its clearance to EDDP (the major quantifiable metabolite) is mediated primarily by hepatic CYP3A4. Both α 1-acid glycoprotein and CYP3A4 are subject to wide between-individual variability, which may be influenced by genetic, environmental, or disease-related factors. Recent population pharmacokinetic analyses have examined factors that may be predictive of methadone oral clearance in maintenance patients. As a surrogate for the unbound fraction of drug, plasma α 1-acid glycoprotein concentration has been shown to have relatively minor predictive value for the oral clearance of methadone, presumably due to between-individual variability in the expression of α 1-acid glycoprotein variants to which methadone binds.
In contrast, CYP3A4 activity, as measured by midazolam oral clearance, explains 22% of the between-individual variability in the oral clearance of unbound (R)-methadone.For many drugs, there is a clearer relationship between plasma concentration and effect than that for dose and effect. Plasma methadone concentration–effect relationships have been reported for postoperative analgesia –, cancer pain , chronic noncancer pain –, experimental pain , sedation , miosis , , withdrawal , subjective opioid effects and mood disturbance. Large between-subject –, but lower within-subject variability for methadone concentrations to achieve analgesia have been reported.
The magnitude of between- and within-subject variability, and how they compare, has not been studied in MMT participants for withdrawal severity, subjective opioid effects, total mood disturbance, miosis, respiratory rate and response to experimentally induced pain. Furthermore, between- and within-subject variability of methadone pharmacokinetics has received limited attention.It is likely that nonholders are at greater risk than holders of seeking illicit opioids or of leaving the MMT programme altogether ,.
Nonholders might be better served with alternative treatments , such as twice-daily methadone, l-α-acetylmethadol (LAAM) , slow release oral morphine or buprenorphine. However, before such strategies are considered, it is important to determine if the pharmacokinetic and pharmacodynamic characteristics of an MMT participant remain relatively constant over time. For example, if withdrawal and total mood disturbance scores are high on one occasion (consistent with nonholding status), would similar results be expected on future occasions? We postulated that, whereas there would be considerable differences between individuals, each individual would show some degree of consistency in methadone disposition and response for a range of pharmacokinetic parameters and pharmacodynamic measures. To test this hypothesis, we compared the between- and within-subject variability in a group of nonholder MMT participants.
PatientsEthical approval was obtained from the Royal Adelaide Hospital Research Ethics Committee. Six self-reported nonholder MMT patients (one female, five male) gave written informed consent to participate. Each had been enrolled in the South Australian Public Methadone Program for at least 6 months prior to entry into the study and had no dose change greater than 15 mg during this period. Demographic details were: age: 21–46 years; weight: 45–107 kg; once daily methadone dose for each subject: subject 1: 20 mg; subject 2: 65 mg; subject 3: 140 mg; subject 4: 105 mg; subject 5: 170 mg; subject 6: 160 mg; comedications (prescribed and illicit) remained unchanged throughout the entire study period and included: benzodiazepines (four subjects); antidepressants (four subjects); cannabis (three subjects); one subject took simvastatin, quinine and naproxen; all smoked tobacco; and only one drank alcohol (less than 20 g ethanol daily).
None had illicit opiates in the urine screen. Subjects were excluded from the study if they were pregnant and if they had positive HIV serology.
All subjects received AUS$250 for completing the three study occasions. Procedures and measuresEach subject was admitted to an inpatient facility for 24 h on three separate occasions. The shortest interval between test visits was 7 days and the longest 16 days.
Subjects arrived at a prearranged time before taking their usual scheduled methadone dose. A urine specimen was obtained to check for illicit drug use. An 18-gauge intravenous catheter (Insyte™, BD Medical, UT, USA) was inserted into the best available arm vein, following which the first 5 mL blood sample was collected. Several procedures were then carried out.Subjective measures included the following: (i) the Methadone Symptoms Checklist (MSC), a self-reported questionnaire that includes three groups of 16 items each, indicating direct opioid effects, opioid withdrawal symptoms, and symptoms which may be characteristic of both direct effects and withdrawal. From this, a withdrawal score, with a maximum value of 16, is obtained ; (ii) the Morphine Benzedrine Group (MBG) Scale of the Addiction Research Centre Inventory which includes 16 items, each of which requires a yes/no response.
Evidence suggests that this is a valid and reliable self-report measure of positive opioid effects; (iii) Profile of Mood States (POMS) which is divided into six empirically derived subscales (anger, depression, confusion, fatigue, tension and vigour) that reflect distinct types and qualities of identifiable affective states. The Total Mood Disturbance (TMD) score (maximum 168) is derived by summing the scores from the subscales. Plasma (R)- and (S)- methadone concentration analysisQuantification of the enantiomers of methadone in plasma was achieved using a previously validated stereoselective HPLC assay. The assay was accurate and reproducible with low (LQC, 54 ng ml −1), medium (MQC, 90 ng ml −1) and high (HQC, 350 ng ml −1) quality control samples for (R)- and (S)-methadone. Inter-assay accuracy and precision (mean accuracy ± precision) were 106 ± 7% (LQC), 103 ± 4% (MQC) and 100 ± 4% (HQC) for (R)-methadone, and 103 ± 7% (LQC), 105 ± 5% (MQC) and 103 ± 6% (HQC) for (S)-methadone. Similarly, intra-assay accuracy and precision were 108 ± 8% (LQC), 106 ± 7% (MQC) and 107 ± 4% (HQC) for (R)-methadone, and 104 ± 7% (LQC), 106 ± 6% (MQC) and 112 ± 5% (HQC) for (S)-methadone. The assay was both precise and accurate at the limit of quantification (15 ng ml −1), with inter-assay accuracy and precision being 101 ± 4% and 104 ± 4% for (R)- and (S)-methadone, respectively.
There were no interfering peaks in any of the chromatograms except in the plasma of subject 5, whose data were excluded from the pharmacokinetic analysis, as quantification of methadone was not possible. Data analysesThe area under the plasma concentration vs. Time curve (AUC) from 0 to 24 h (the interdosing interval) was calculated by the linear trapezoidal method. The apparent oral clearance (CL/ F) was calculated from the expression CL/ F = dose/AUC, where F is the oral bioavailability. To allow for comparisons across subjects, all calculations were based on (R)- or (S)-methadone concentrations normalized to 35 mg of each enantiomer (70 mg rac-methadone). Estimates of the dose-corrected maximum and minimum plasma concentrations ( C max and C min) were obtained by visual inspection of the data.For all subjects, a significant fluctuation in the pharmacodynamic measures was observed across the 24 h observation period. Thus, for each measure, the data were expressed as the area under the end-point vs.
Time curve (AUC) from 0 to 24 h, calculated by the trapezoidal method. Statistical methodsAn exploratory analysis was initially conducted by viewing a scatterplot of each measure vs. Subject (for each variable), with an indicator for the order in which the measurements were taken. This allowed for a visual determination of whether there was an order effect on the measurements. In addition, these scatterplots allowed for a visual determination of the extent to which the variability of each variable changed proportionally with the magnitude of the values measured for each variable. If a proportional relationship was not evident, then the coefficient of variation was not a justified summary measure. As a result, a visual inspection of scatterplots led to the determination of an appropriate summary measure.An analysis of variance ( anova) with mixed effects was conducted for each variable.
Subject effects were considered as random as the focus of the analysis was not on the differences between individual subjects, but on the reasons for this variation. Order was included initially as a fixed effect in an anova for all variables (alongside a random subject effect). If this fixed effect was found to be significant it was retained in the model; otherwise it was removed.To test the null hypothesis that the random effect for between-subject variation was equal to zero, we constructed an F statistic, expressed as: where is the estimated variation between individuals and is the estimated variation within individuals (or residual variation).The probability of obtaining an F statistic like this (or more extreme) from our data, if the null hypothesis was true was then determined for each variable. F statistics were compared with an F distribution with (5,12) degrees of freedom for all anovas, except that for those associated with the analysis of plasma (R)-methadone and (S)-methadone concentration-related data, where there were (4,10) degrees of freedom.If the P-value obtained was less than 0.05, we concluded that there was statistically significant evidence against the null hypothesis. In this case we then constructed our best estimate of the ratio of variances, expressed as:. Alternatively, if the P-value obtained was greater than 0.05, we accepted that there was no statistically significant evidence against the null hypothesis, and the estimated ratio was not relevant.In addition, if the null hypothesis was rejected, an approximate 95% confidence interval for measures within subjects was constructed about the grand mean using the 95% margin of error, i.e. For each variable.
Exploratory statistical analysisVisual inspection of scatterplots of measures vs. Subject (for each variable) suggested that there was no order effect, with the exception of (R)-methadone AUC. In addition, with the possible exception of (R)-methadone CL/F, and (R)- and (S)-methadone C min, variability did not appear to increase proportionally with the magnitude of the measure, indicating that the ratio of variance estimates is a more appropriate summary measure for the majority of variables.
However, even for these possible exceptions, it was not convincing that a coefficient of variation was entirely appropriate. As a consequence, estimating between- and within-subject variability using a coefficient of variation could afford a misleading interpretation of the comparative variability.In an exploratory analysis, anovas with mixed effects were performed for all variables including a random effect for subject and a fixed effect for order.
The results for all analyses suggested that order was statistically nonsignificant, with the exception of those for (R)-methadone C max and (R)-methadone CL/ F. Consequently, in the final analyses for these variables, the fixed effect of order was retained and for all others it was removed. In all cases there was evidence that the random effect for subject differences was statistically significantly different from zero. ResultsValues for all of the variables examined, subjects were relatively consistent between occasions , with the possible exception of (S)-methadone CL/ F and direct opioid effects.
In contrast, there was much greater between-subject variability, as is evident from the best variance ratio estimate.shows the relatively small within-subject and much greater between-subject variability in the CL/F for (R)-methadone for the five individuals in whom this could be measured. Between-subject variability was significantly greater (up to nearly 8-fold) than within-subject variability for all pharmacokinetic parameters. The smallest variance ratio was obtained for C max, and was similar for both (R)-methadone (1.5) and (S)-methadone (1.3). (R)-methadone displayed consistently less within-subject variability in all pharmacokinetic parameters (. (R)-methadone apparent oral clearance (CL/F) in five MMT subjects on three separate occasions.
The daily rac-methadone doses were 20 mg (subject 1), 65 mg (subject 2), 140 mg (subject 3), 105 mg (subject 4), and 160 mg (subject 6)The withdrawal score AUC for all individuals on each occasion is shown in. The results indicate that there is relatively small within-subject variability for all patients, except subject 4, whose AUC varied approximately 4-fold over the three occasions. There also appears to be little relationship between the daily racemic methadone dose, and the withdrawal score AUC. For example, patient 3, whose daily dose was 140 mg, and patient 6 whose daily dose was 160 mg, exhibited the highest withdrawal score AUC values. Furthermore, it is clear that the variation does not appear to increase as the magnitude of the score increases. This illustrates why a coefficient of variation is not an appropriate summary measure in this context, and indices of variance are much more informative. Between-subject variability was significantly ( P.
DiscussionTo our knowledge, this is the first study to have investigated within-subject and between-subject variability in the pharmacokinetics and pharmacodynamics of methadone at steady state in patients on MMT. To enable a comparison of these estimates of variability, repeated measures on several occasions must be obtained.
On each occasion, subject adherence (compliance) with the dosing regimen must be complete, and drug dose and extrinsic factors governing bioavailability and clearance must remain unchanged. Our study design fulfilled these criteria. We chose nonholders because they are of particular clinical interest due to their potentially greater risk of not remaining in treatment.The coefficient of variation is a popular summary measure of variation in experimental data, and assumes a constant proportional relationship between the standard deviation and the magnitude of the measure. Whereas this is often the case, for example in drug analysis, problems arise when there is evidence that this underlying assumption is not met, as was the case with our data.
Thus, we report the between- to within-individual variance ratio, which affords a much more appropriate, reliable and informative index of variation. However, this ratio does not provide a measure of the magnitude of within-subject variability.
Such information is afforded by the margin of error for the 95% confidence interval for within-subject measures. In theory, the margin of error can be applied directly to the value of any variable, as this measure is independent of the magnitude of the variable. Therefore, to provide a context for the margin of error, we calculated 95% confidence intervals using the grand mean for all variables (i.e. A ‘typical value’).
The degree of within-subject variability can then be assessed by inspecting the width of the 95% confidence intervals.As postulated, within-subject variability in all end points was substantially less than between-subject variability. The latter for several pharmacokinetic parameters has been reported for patients in MMT programmes since the mid 1970s. However, most of these reports have dealt with the racemic compound and not the individual enantiomers. Values for between-subject variability at steady state in MMT patients for clearance have ranged from 1.5-fold to 5-fold for the racemate; 3.5-fold for the (R)-enantiomer and about 5-fold for the (S)-enantiomer. Similar data have been reported in single dose studies in healthy subjects, chronic pain patients and in opioid users. In a recently published population pharmacokinetic analysis in a large cohort, we reported a 7-fold range of values, with a 40% coefficient of variation for intersubject variability in (R)-methadone apparent oral clearance. The range of such values reported here is consistent with previous literature, and similar variability was found for the other pharmacokinetic parameters.There are few reports regarding within-subject variability in methadone pharmacokinetics.
Eap and coworkers compared the between-subject variability (about 5-fold) and within-subject variability (negligible) in the ratio of plasma (R)- to (S)-methadone concentrations in ‘trough’ blood samples from 20 MMT patients. Although these data provide evidence that the influence of stereoselectivity on the disposition of methadone is consistent in an individual, it does not afford any indication of the within-subject variability in methadone blood concentrations, and hence methadone clearance. In contrast, our analyses show that between-subject variability in methadone pharmacokinetic parameters is up to 8-fold greater than within subject variability. Importantly, the apparent oral clearance of the active (R)-isomer appears to be relatively consistent between occasions within an individual, as the within-subject 95% CI ranged only 1.7-fold at the mean clearance value. In contrast (S)-methadone shows greater within-subject variability (95% CI ranged 3.4-fold), which may explain the smaller between- to within-subject variance ratio estimate for (S)-methadone (2.7) compared with (R)-methadone (4.9). This may be of clinical importance as high (S)-methadone concentrations may be associated with significant adverse events.Intuitively one might predict that the between-individual variability in drug disposition is greater than the within-individual variability.
Examples include: alcohol , gabapentin , talinolol , ropivacaine , hydroxychloroquine and etoposide. It is noteworthy that there are no common features determining the disposition mechanisms for these examples.There are other drugs, however, for which within-subject variability in drug disposition is not greatly different from between-subject variability. Examples include avitriptan , verapamil , , frusemide and ibuprofen. Our results for methadone, and those listed above, indicate that specific, well designed trials need to be conducted with each drug to determine differences in between- and within-subject variability in disposition.In contrast to the variability reported for its pharmacokinetics, variability in the pharmacodynamics of methadone has been much less frequently studied. Boulton and coworkers reported a 40% between-subject coefficient of variation in pupil response following a single dose of methadone in eight healthy subjects.
In a cohort of MMT patients, Dyer and coworkers found a coefficient of variation of 75% for between-subject variability in the EC 50 for pupil diameter ; a 25% value for total mood disturbance ; a 48% value for direct opioid effects ; and a 97% value for withdrawal score. Inturrisi and coworkers reported a 10-fold to 30-fold between-subject variability in the EC 50 for pain relief in patients with cancer being treated with a variety of opioids. One subject was studied twice over an interval of 2.5 months ; and the EC 50 values on the two occasions were almost identical. Our results demonstrate that objective measures of opioid effect appear to be consistent within an individual, relative to a much greater between-subject variability. In contrast, subjective opioid effects appear to be more variable between occasions, which may explain the smaller between- to within-variance ratio estimates for subjective measures.Despite the small numbers in this study, we have been able to show highly significant differences in the between- to within-subject variability for all of the pharmacokinetic and pharmacodynamic end points measured.
Importantly, this includes both the pharmacokinetic parameters, and both objective (such as pupil diameter) and subjective (such as withdrawal) pharmacodynamic measures. There is a reasonable understanding of factors that might influence variability in methadone disposition. These include hepatic CYP3A4 activity and the presence of interacting comedications. In contrast, factors influencing variability in pharmacodynamics are poorly defined.
However, it is likely that pharmacogenomics play a central role, as methadone is a p-glycoprotein substrate, genetic variations of which will affect the brain distribution kinetics of methadone, and variations in the genes for the µ opioid receptor, its G-proteins and transduction elements contribute to variability in the response to opioid drugs. Despite this, we have shown a striking difference in the between- to within-subject variability in both subjective measures, such as withdrawal, positive opioid effects and pain sensation, and more objective measures such as pupil diameter and respiration rate. This suggests that, in this population, these end-points are likely to provide a clinically useful guide to treatment.Four subjects were receiving daily methadone doses in excess of 100 mg. This observation is consistent with our previously reported findings that many nonholders are taking relatively high doses of methadone ,.
As a result, our data suggest that a nonholder taking a relatively high dose on one occasion is more likely than not to behave as a nonholder on subsequent occasions. We predict that the same would apply to a holder, although this would need to be confirmed.Our findings have several clinical implications. If therapeutic drug monitoring is employed and blood is collected just before the next dose (C min), the clinician can be reassured that the result will be reasonably consistent from occasion to occasion over a 1–2 month period, as long as the dose and adherence remain unchanged. Our data suggest that an individual experiences fairly consistent pharmacodynamic effects from occasion to occasion on a constant treatment regimen. This is especially the case for objective effects, and the subjective effects, which provide an index of mood disturbance, pain tolerance and, most importantly, withdrawal severity.
Therefore, identification of holder/nonholder status early in the course of MMT might influence the choice of the most appropriate substitution drug. In this case, an alternative to methadone, such as buprenorphine , or slow release oral morphine might be more appropriate. This may translate into improved retention in maintenance programs and a greater likelihood of a better clinical outcome. We thank Dr Kyle Dyer for advice during the early phase of the study, and the staff of the Warinilla Clinic of the Drug and Alcohol Services Council of South Australia for helping to recruit the subjects and for their care during their inpatient stay. We thank the National Health & Medical Research Council of Australia for financial support (Grant # 990586).None of the authors have any financial or personal relationships that could potentially be perceived as influencing the research described in this manuscript.
Mixed ANOVA using SPSS Statistics IntroductionA mixed ANOVA compares the mean differences between groups that have been split on two 'factors' (also known as independent variables), where one factor is a 'within-subjects' factor and the other factor is a 'between-subjects' factor. For example, a mixed ANOVA is often used in studies where you have measured a dependent variable (e.g., 'back pain' or 'salary') over two or more time points or when all subjects have undergone two or more conditions (i.e., where 'time' or 'conditions' are your 'within-subjects' factor), but also when your subjects have been assigned into two or more separate groups (e.g., based on some characteristic, such as subjects' 'gender' or 'educational level', or when they have undergone different interventions). These groups form your 'between-subjects' factor. The primary purpose of a mixed ANOVA is to understand if there is an interaction between these two factors on the dependent variable. Before discussing this further, take a look at the examples below, which illustrate the three more common types of study design where a mixed ANOVA is used. Study Design #1. Study Design #2.
Study Design #3Study Design #1Your within-subjects factor is time.Your between-subjects factor consists of conditions (also known as treatments).Imagine that a health researcher wants to help suffers of chronic back pain reduce their pain levels. The researcher wants to find out whether one of two different treatments is more effective at reducing pain levels over time. Therefore, the dependent variable is 'back pain', whilst the within-subjects factor is 'time' and the between-subjects factor is 'conditions'. More specifically, the two different treatments, which are known as 'conditions', are a 'massage programme' (treatment A) and 'acupuncture programme' (treatment B). These two treatments reflect the two groups of the 'between-subjects' factor.In total, 60 participants take part in the experiment.
Of these 60 participants, 30 are randomly assigned to undergo treatment A (the massage programme) and the other 30 receive treatment B (the acupuncture programme). Both treatment programmes last 8 weeks.
As mentioned above, the primary purpose of a mixed ANOVA is to understand if there is an interaction between your within-subjects factor and between-subjects factor on the dependent variable. Once you have established whether there is a statistically significant interaction, there are a number of different approaches to following up the result. In particular, it is important to realize that the mixed ANOVA is an omnibus test statistic and cannot tell you which specific groups within each factor were significantly different from each other. For example, if one of your factors (e.g., 'time') has three groups (e.g., the three groups are your three time points: 'time point 1', 'time point 2' and 'time point 3'), the mixed ANOVA result cannot tell you whether the values on the dependent variable were different for one group (e.g., 'Time point 1') compared with another group (e.g., 'Time point 2'). It only tells you that at least two of the three groups were different. Since you may have three, four, five or more groups in your study design, as well as two factors, determining which of these groups differ from each other is important.
You can do this using post hoc tests, which we discuss later in this guide. In addition, where statistically significant interactions are found, you need to determine whether there are any 'simple main effects', and if there are, what these effects are (again, we discuss this later in our guide).If you are unsure whether a mixed ANOVA is appropriate, you may also want to consider how it differs from a two-way repeated measures ANOVA.
Both the mixed ANOVA and two-way repeated measures ANOVA involve two factors, as well as a desire to understand whether there is an interaction between these two factors on the dependent variable. However, the fundamental difference is that a two-way repeated measures ANOVA has two 'within-subjects' factors, whereas a mixed ANOVA has only one 'within-subjects' factor because the other factor is a 'between-subjects' factor. Therefore, in a two-way repeated measures ANOVA, all subjects undergo all conditions (e.g., if the study has two conditions – a control and a treatment – all subjects take part in both the control and the treatment). Therefore, unlike the mixed ANOVA, subjects are not separated into different groups based on some 'between-subjects' factor (e.g., a characteristic such as subjects' 'gender' or 'educational level', or so that they only receive one 'condition': either the control or the treatment). Therefore, if you think that the mixed ANOVA is not the test you are looking for, you may want to consider a. Alternately, if neither of these are appropriate, you can use our, which is part of our enhanced content, to determine which test is appropriate for your study design.In this 'quick start' guide, we show you how to carry out a mixed ANOVA with post hoc tests using SPSS Statistics, as well as the steps you will need to go through to interpret the results from this test. However, before we introduce you to this procedure, you need to understand the different assumptions that your data must meet in order for a mixed ANOVA to give you a valid result.
We discuss these assumptions next. SPSS Statistics AssumptionsWhen you choose to analyse your data using a mixed ANOVA, much of the process involves checking to make sure that the data you want to analyse can actually be analysed using a mixed ANOVA.
You need to do this because it is only appropriate to use a mixed ANOVA if your data 'passes' seven assumptions that are required for a mixed ANOVA to give you a valid result. In practice, checking for these assumptions requires you to use SPSS Statistics to carry out a few more tests, as well as think a little bit more about your data. Whilst it is not a difficult task, it will take up most of your time when carrying out a mixed ANOVA.Before we introduce you to these seven assumptions, do not be surprised if, when analysing your own data using SPSS Statistics, one or more of these assumptions is violated (i.e., not met). This is not uncommon when working with real-world data rather than textbook examples. However, even when your data fails certain assumptions, there is often a solution to try and overcome this. First, let’s take a look at these seven assumptions:. Assumption #1: Your dependent variable should be measured at the continuous level (i.e., they are either interval or ratio variables).
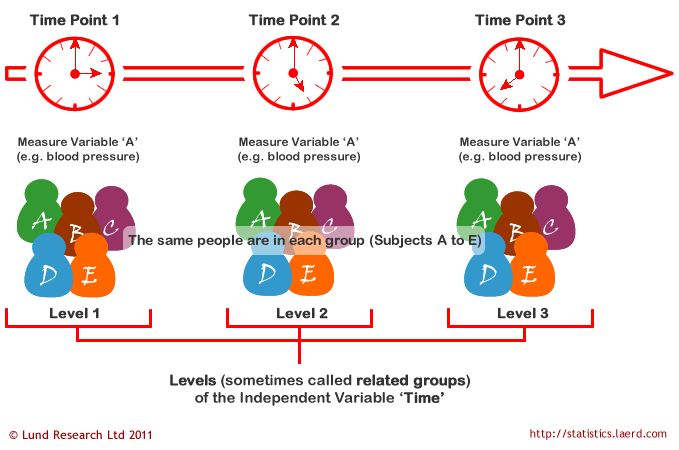
Examples of continuous variables include revision time (measured in hours), intelligence (measured using IQ score), exam performance (measured from 0 to 100), weight (measured in kg), and so forth. You can learn more about interval and ratio variables in our article:. Assumption #2: Your within-subjects factor (i.e., within-subjects independent variable) should consist of at least two categorical, 'related groups' or 'matched pairs'. 'Related groups' indicates that the same subjects are present in both groups.
The reason that it is possible to have the same subjects in each group is because each subject has been measured on two occasions on the same dependent variable, whether this is at two different 'time points' or having undergone two different 'conditions'. For example, you might have measured 10 individuals' performance in a spelling test (the dependent variable) before and after they underwent a new form of computerized teaching method to improve spelling (i.e., two different 'time points'). You would like to know if the computer training improved their spelling performance. The first related group consists of the subjects at the beginning of the experiment, prior to the computerized spelling training, and the second related group consists of the same subjects, but now at the end of the computerized training.
Assumption #3: Your between-subjects factor (i.e., between-subjects factor independent variable) should each consist of at least two categorical, 'independent groups'. Example independent variables that meet this criterion include gender (2 groups: male or female), ethnicity (3 groups: Caucasian, African American and Hispanic), physical activity level (4 groups: sedentary, low, moderate and high), profession (5 groups: surgeon, doctor, nurse, dentist, therapist), and so forth.
Assumption #4: There should be no significant outliers in any group of your within-subjects factor or between-subjects factor. Outliers are simply single data points within your data that do not follow the usual pattern (e.g., in a study of 100 students' IQ scores, where the mean score was 108 with only a small variation between students, one student had a score of 156, which is very unusual, and may even put her in the top 1% of IQ scores globally). The problem with outliers is that they can have a negative effect on the mixed ANOVA, distorting the differences between the related groups (whether increasing or decreasing the scores on the dependent variable), which reduces the accuracy of your results. Fortunately, when using SPSS Statistics to run a mixed ANOVA on your data, you can easily detect possible outliers. In our enhanced mixed ANOVA guide, we: (a) show you how to detect outliers using SPSS Statistics, whether you check for outliers in your 'actual data' or using 'studentized residuals'; and (b) discuss some of the options you have in order to deal with outliers. Assumption #5: Your dependent variable should be approximately normally distributed for each combination of the groups of your two factors (i.e., your within-subjects factor and between-subjects factor).
Whilst this sounds a little tricky, it is easily tested for using SPSS Statistics. Also, when we talk about the mixed only requiring approximately normal data, this is because it is quite 'robust' to violations of normality, meaning that assumption can be a little violated and still provide valid results. You can test for normality using, for example, the Shapiro-Wilk test of normality (for 'actual data') or Q-Q Plots (for 'studentized residuals'), both of which are simple procedures in SPSS Statistics. In addition to showing you how to do this in our enhanced mixed ANOVA guide, we also explain what you can do if your data fails this assumption (i.e., if it fails it more than a little bit).
Assumption #6: There needs to be homogeneity of variances for each combination of the groups of your two factors (i.e., your within-subjects factor and between-subjects factor). Again, whilst this sounds a little tricky, you can easily test this assumption in SPSS Statistics using Levene’s test for homogeneity of variances. In our enhanced mixed ANOVA guide, we (a) show you how to perform Levene’s test for homogeneity of variances in SPSS Statistics, (b) explain some of the things you will need to consider when interpreting your data, and (c) present possible ways to continue with your analysis if your data fails to meet this assumption. Assumption #7: Known as sphericity, the variances of the differences between the related groups of the within-subject factor for all groups of the between-subjects factor (i.e., your within-subjects factor and between-subjects factor) must be equal.
Fortunately, SPSS Statistics makes it easy to test whether your data has met or failed this assumption. Therefore, in our enhanced mixed ANOVA guide, we (a) show you how to perform Mauchly's Test of Sphericity in SPSS Statistics, (b) explain some of the things you will need to consider when interpreting your data, and (c) present possible ways to continue with your analysis if your data fails to meet this assumption.You can check assumptions #4, #5, #6 and #7 using SPSS Statistics. Just remember that if you do not run the statistical tests on these assumptions correctly, the results you get when running a mixed ANOVA might not be valid. This is why we dedicate a number of sections in our enhanced guide to help you get this right.
You can find out about our enhanced content as a whole, or more specifically, learn how we help with testing assumptions.In the section, we illustrate the SPSS Statistics procedure that you can use to carry out a mixed ANOVA on your data. First, we introduce the example that is used in this guide. SPSS Statistics ExampleA researcher wanted to discover whether the intensity of an exercise-training programme, but with equal calorific expenditure, had an effect on cholesterol concentration over a six-month period. Therefore, the dependent variable was 'cholesterol concentration', the within-subjects factor was 'time' and the between-subjects factor was the 'conditions' (N.B., each of these variables are explained further below).To answer this, 60 participants were recruited to take part in the experiment, randomly split into three each groups of 20 participants. Each of these three groups of 20 participants received a different 'condition': in one group, participants did not change their current sedentary lifestyle (i.e., this was Group #1, also called the 'control' group); in another group, participants underwent a low-intensity exercise-training programme that expended 1000 kCal per week (i.e., this was Group #2, also called 'treatment A'); the final group underwent a high-intensity exercise-training programme that also expended 1000 kCal per week, but therefore exercised for less total time (i.e., this was Group #3, also called 'treatment B'). All of the conditions (i.e., the control, treatment A and treatment B) lasted six months.
During this period, the dependent variable, 'cholesterol concentration', was measured three times: 'at the beginning of the experiment' (time point #1), 'mid-way through the six months' (time point #2) and 'at the end of the experiment' (time point #3). These three time points (i.e., time point #1, time point #2 and time point #3) represent the three groups of the within-subjects factor, 'time'. SPSS Statistics Setup in SPSS StatisticsIn this example, there are three variables: (1) the dependent variable, cholesterol, which is the cholesterol concentration (in mmol/L); (2) the between-subjects factor, group, which has three categories: 'Control' (control group), 'Int1' (treatment A) and 'Int2' (treatment B); and (3) the within-subjects factor, time, which has three categories: 'pre', 'mid' and 'post'.Participants' cholesterol concentration was recorded in the variable pre for pre-intervention, mid for mid-way through and post for post-intervention. These three variables make up the within-subjects factor, time, and the scores within these three variables reflect the dependent variable, cholesterol. The different interventions were stored in the variable, group, where 'Control' is the control group, 'Int1' is the low-intensity training intervention, and 'Int2' is the high-intensity training intervention. In variable terms, the researcher wishes to know if there is an interaction between group and time on cholesterol.In our enhanced mixed ANOVA guide, we show you how to correctly enter data in SPSS Statistics to run a mixed ANOVA. You can learn about our enhanced data setup content.
Alternately, we have a generic, 'quick start' guide to show you how to enter data into SPSS Statistics, available. SPSS Statistics Test Procedure in SPSS StatisticsThe 20 steps below show you how to analyse your data using a mixed ANOVA in SPSS Statistics, including which post hoc test to select to determine where any differences lie, when none of the seven assumptions in the previous section, have been violated. At the end of these 20 steps, we explain what results you will need to interpret from your mixed ANOVA.
If you are looking for help to make sure your data meets assumptions #4, #5, #6 and #7, which are required when using a mixed ANOVA and can be tested using SPSS Statistics, we show you how to do this in our enhanced content (see ).Click Analyze General Linear Model Repeated Measures. On the top menu, as shown below. Published with written permission from SPSS Statistics, IBM Corporation.Transfer time and ' group.time' (the interaction term) from the Factor(s) and Factor Interactions: box to the Display Means For: box by highlighting them and clicking the button. This will activate the C ompare main effects checkbox (i.e., it will no longer be greyed out). Tick this checkbox and select 'Bonferroni' from the drop-down menu under Co nfidence interval adjustment. Then, in the –Display– area, tick the Descriptive statistics, Estimates of effect size and Homogeneity tests checkboxes.
After you have done all this, you will be presented with the following screen. SPSS Statistics Analysing the Output from a Mixed ANOVAThe output generated by SPSS Statistics is quite extensive and can provide a lot of information about your analysis. However, if there was a statistically significant interaction between your two factors on the dependent variable, you will need to carry out some additional steps in SPSS Statistics. Below we briefly explain the main steps that you will need to follow to interpret your mixed ANOVA results, and where required, perform additional analysis in SPSS Statistics.
If you want to know how to go through all these sections step-by-step, together with the relevant SPSS Statistics output, we show you how to do this in our enhanced mixed ANOVA guide. You can learn more about our enhanced content in general. First, take a look through these steps:. Step #1: You need to interpret the results from your assumption tests to make sure that you can use a mixed ANOVA to analyse your data.
